Your Cart is Empty
Free shipping on US orders! 20% off your first order | Code: SkyGenius20
Free shipping on US orders! 20% off your first order | Code: SkyGenius20
Free shipping on US orders! 20% off your first order | Code: SkyGenius20
Free shipping on US orders! 20% off your first order | Code: SkyGenius20
by Genius Sky May 14, 2021 14 min read

Quick Links
Just like other lithium-ion cells, the 18650 battery is named after dimensions.
The first two numbers(18) refer to the diameter of the battery in millimeters.
The next two numbers (65) stand for the height, and the final number (0) means the shape of the battery.

In this case:
Voltage, or electrical pressure, is the difference in electric potential between two points.
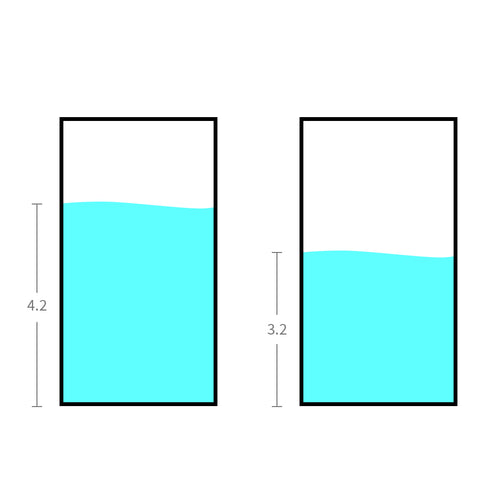
We can think of the 18650 battery as a bucket of water.
Water level, indicating the height of the water, is somehow very similar to voltage.
Because they are both used to gauge the range, difference between the lowest and highest.
When the water is consumed, the water level declines. After refilling, the water level rises.
So you know that voltage decreases when the battery is running/discharging, and increases again when the battery is being charged.
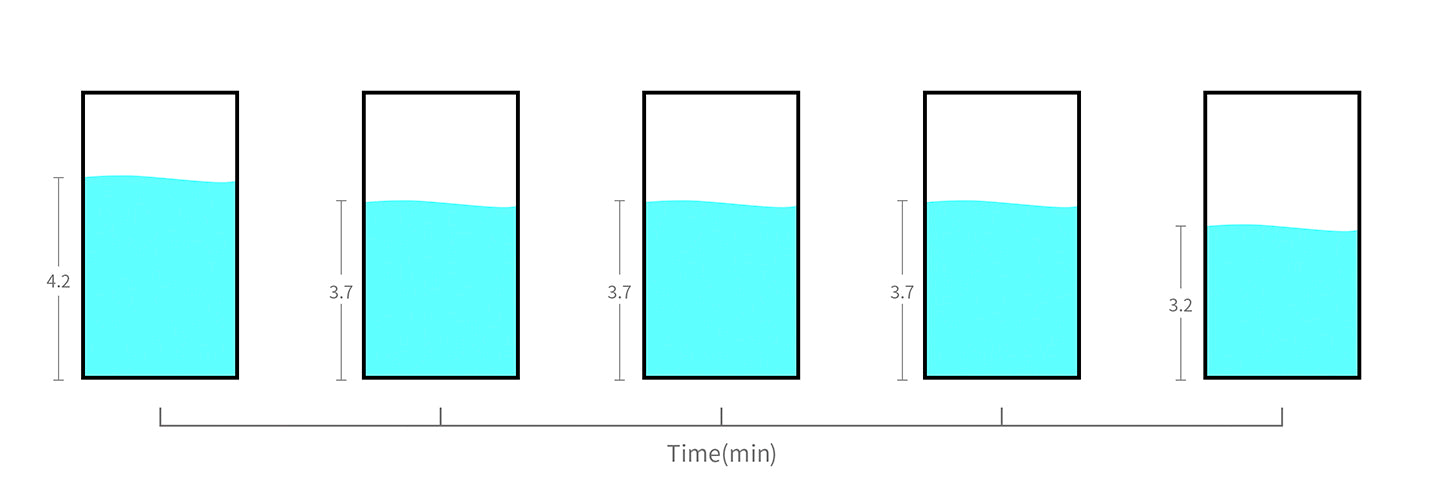
The voltage of 18650 lithium battery doesn't decrease evenly.
It begins at 4.2V and decreases slightly. It then drops rapidly to around 3.2V, until it stops working altogether.
From the graph, you can see that most of the time, it remains at around 3.7V midway through the discharging cycle.
Nominal voltage is often the midpoint of that range.
Cell manufacturers mark the nominal voltage of their lithium-ion as 3.6V or 3.7V.
For marketing purposes, some manufactures would mark it as 3.7V on the package to imply that it's more powerful.
You may wonder, why the nominal voltage of a 18650 battery has to be 3.6V or 3.7V.
Why isn't it 1.1V, 2.3V, or any other arbitrary number?
The nominal voltage is a function of anode and cathode materials, as well as impedance.
This sentence sounds complicated to understand.
Let's put it this way.
Nominal voltage depends on the chemistry of the battery.
Battery chemistry and their nominal voltage
| Chemistry | Nominal voltage |
| Six-cell lead acid | 12.6V |
| Lithium-ion | 3.7V |
| Alkaline or Zinc-carbon | 1.5V |
| NiMH or NiCd | 1.2V |
For lithium-ion batteries, the mid-way point is between 3.6V to 3.7V.
This value varies among other types of batteries.
You have a 3.7V 2600 mAh 18650 battery cell.
You use it to power a 5V, 3W clip on fan.
However, the 3.7 voltage is not sufficient to power those devices that require 5V or more.
What does this mean?
Here comes the water bucket analogy again.
Let's suppose you want to spin a small waterwheel because you enjoy watching the rotation.
In the figure below, you can see this waterwheel contraption. It has a bucket sitting next to it with a tap halfway down.
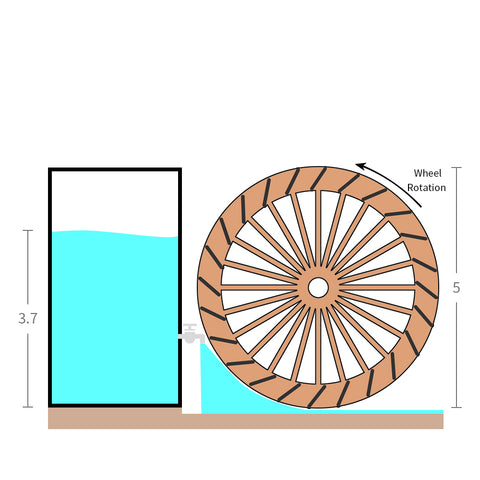
The jet of water doesn't reach a high enough height to push the waterwheel.
We move the same quantity (or volume) of water to a slimmer and taller bucket. This allows the water level to increase.
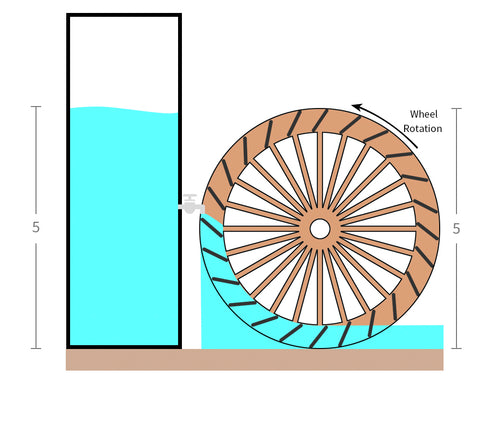
This allows it to work, and the little waterwheel will spin as you wish.
You have a 3.7V 2600mAh battery, and you are expecting it can power a 600mAh fan for 4 hours or more.
2600mAh / 600mAh = 4.3 h
This calculation seems straightforward, but it's not correct.
Let's go back to the water example.
One bucket has a wider base area. The other one is smaller in diameter.
But you can't say the first bucket holds more water depending on the size of the bottom area.
The water depth also counts, right?
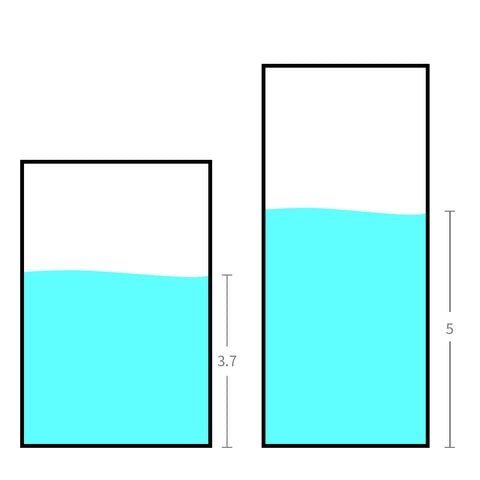
What really matters is the total energy stored in the battery or, in other words, how much water is in the bucket.
So, here comes another unit, Wh.
Wh is short for Watt Hours.
It describes the energy capcity or capacity do useful work stored in the battery.
If you are buying a replacement battery, you might see their capacity listed in mAh (milliamps hours).
"Wait. Didn't you just say Wh stands for battery capacity? How come all the battery capacity was marked in mAh?"
I know you must be confused right now.
Relax, the water bucket analogy will help you sort things out over and over again.
Imagine you are looking to buy a bucket online.
The product page only tells you the radius and height. They think you don't necessarily need to know the volume.
When indicating battery capacity, the relationship between mAh and Wh is very similar to the radius and volume of a bucket.
If you take a closer look, you will find that battery capacity was actually given in 3.7V 2600mAh.
It's like telling you how high(3.7V) and how wide(2600mAh) this product is.
While mAh only suggests "two dimensions" of capacity, Wh is a solid "three dimensions" figure that calculates battery capacity as a whole.
Ah * V = Wh
You may not have heard of AH before.
Don't feel stressed about another unknown terminology.
Ah is just a different unit that is equal to 1000mAh
For a 3.7V 2600mAh battery, the Wh will be:
2600mAh * 3.7V
2.6 Ah * 3.7V = 9.62 Wh
So now we know the energy of a 3.7V 2600mAh battery is 9.62Wh.
Like we said before, Wh is similar to the amount of water in the bucket.
So, what does W mean?
While the term Wh is used to describe the energy stored in a battery, W is more frequently used to describe how much power (power is energy per unit of time) your device consumes.
Different appliances require different amounts of W (Watts or wattage)
Assuming there are 3 small waterwheels, the weight of their bodies differs.
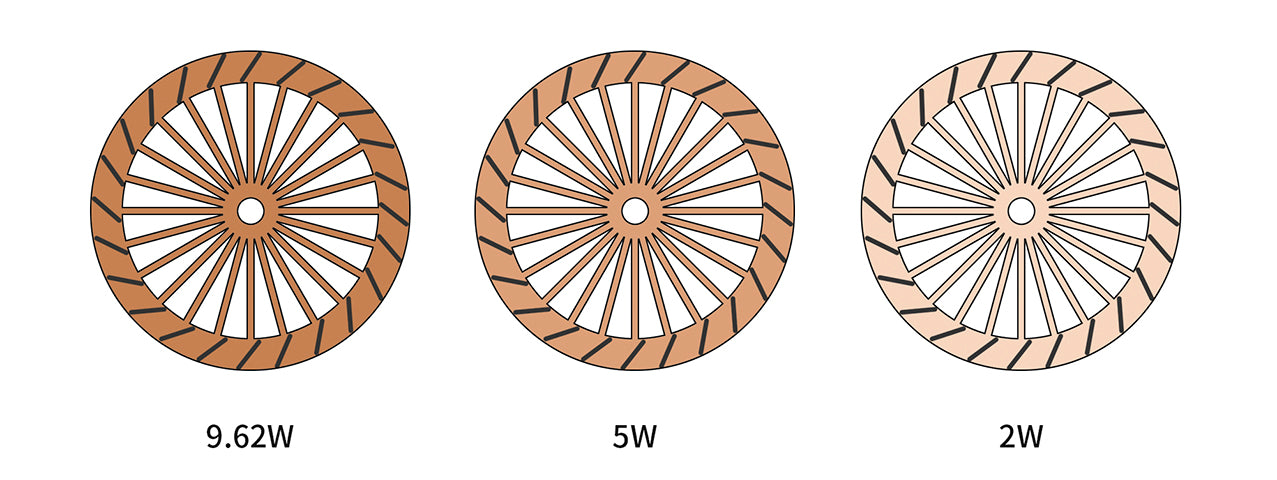
To spin the wheel, the heaviest one requires a strongest jet of water.
The other two wheels are lighter, which means less water is needed to get them to continuously spin.
We can reduce water flow from the tap to prevent any waste.
If we think about the tap pushing out electricity instead of water, we could say it's delivering different numbers of Watts for different water wheels.
Now if the bucket was filled with 9.62 Wh of water, it could supply the heaviest waterwheel for one hour.
We would say this waterwheel consumes 9.62W.
Other waterwheels, however, only consume 5W and 2W.
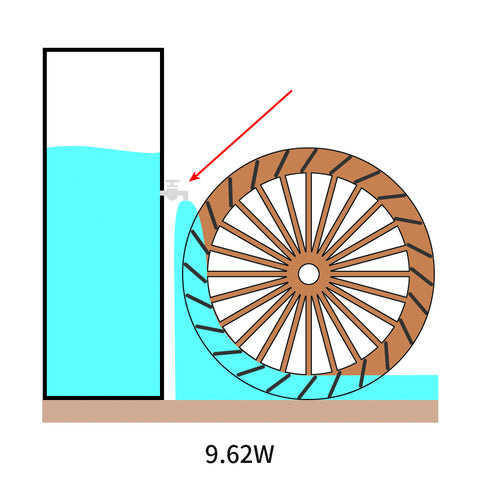
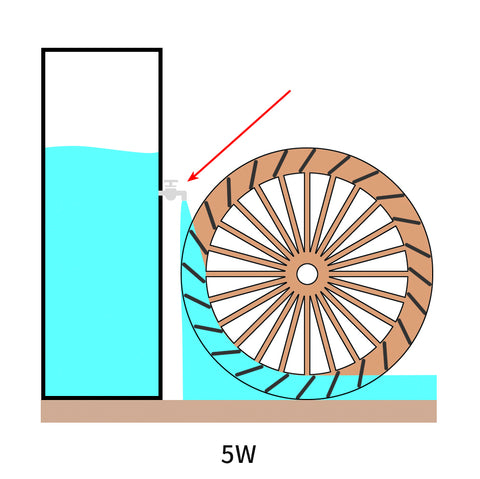
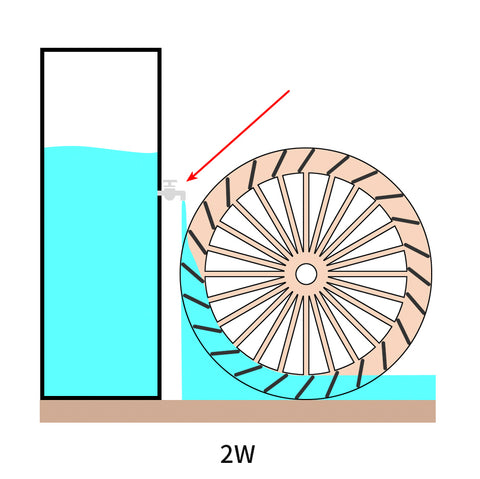
To find out how long these waterwheels keep spinning, just do the math:
9.62Wh / 9.62W = 1h
9.62Wh / 5W = 1.9h
9.62Wh / 2W = 4.8h
Now that you understand the terminologies of voltage, mAh, Wh, and wattage, it's time to calculate the battery runtime.
We have used some examples like a 9.62Wh water bucket can supply the 2W waterwheel for 4.8 hours.
But that's not realistic.
You may have noticed that the water stops coming out when the water is level with the tap.
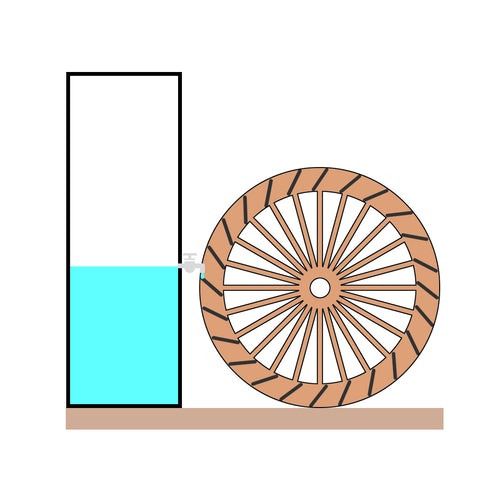
So, the bucket won't completely run out of water.
The same applies to the battery.
It will disconnect the device at a certain voltage.
A 18650 battery is not supposed to run dry, or it will become permanently damaged (dead).
As the amount of water remains the same, if you want to increase the height, you can simply move water from one bucket to another.
When it comes to batteries, you cannot literally change the "container". This is not how a battery works.
In reality, using a 3.7V battery to power a 5V device, a 3.7V to 5V boost converter required.
You can think of it as a pump.
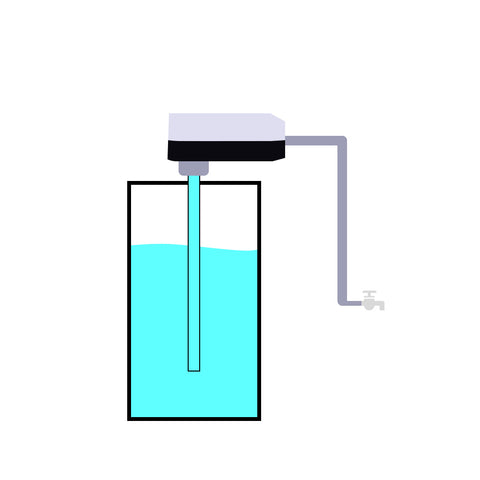
As a working water pump requires energy, the 3.7V to 5V boost converter will consume battery power as well.
So, not all the battery energy is used by the device.
The boost converter also consumes battery energy.
During the conversion from 3.7V to 5V, you lose some battery energy.
mAh is not a direct indicator of the stored energy. The first step is to calculate the battery energy Wh.
For a 3.7V 2600mAh 18650 battery, the Wh will be:
3.7V x 2600mAh
3.7V x 2.6Ah = 9.62Wh

Then, we take the conversion efficiency (assume, typically 65%) into consideration.
9.62Wh x 65% = 6.253Wh
We know the "realistic" battery energy is approximately 6.25 Wh.
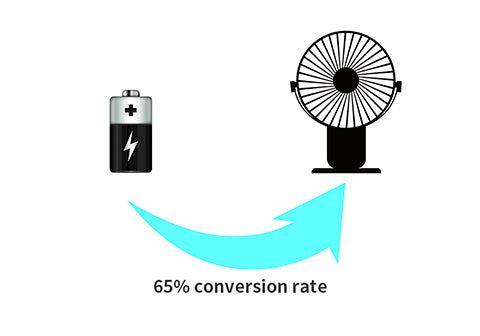
Using the battery to power a 3W device, the runtime will be:
6.253Wh / 3W = 2.08h
That's approximately two hours of runtime.
Sometimes, you don't know the exact wattage of your device. Manufacturers put something like "5V @ 0.6A" on the package instead.
No problem.
Based on the formula of A*V=W, you know the wattage is:
5V x 0.6A =3W
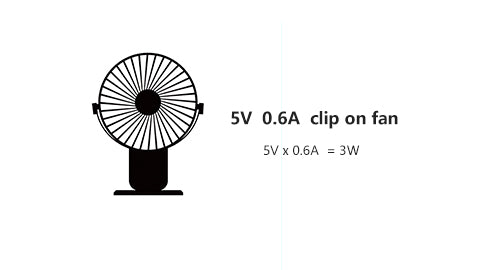
The below graph will help you organize the calculation process.

All batteries have the same three basic parts:
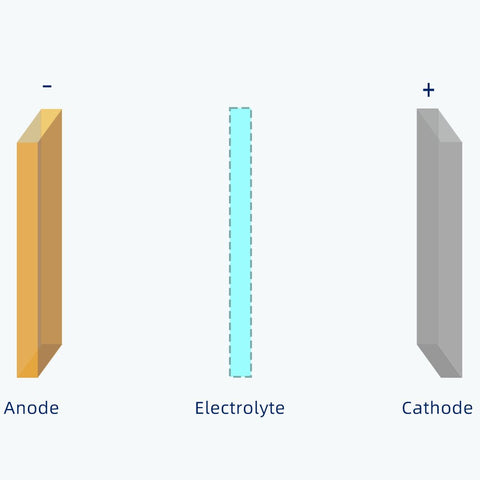
Generally, the positive electrode of 18650 cell is a metal oxide that contains lithium atoms.
The negative electrode is typically made from carbon, which has a layered structure, called graphite.
These layers are loosely bonded, so, lithium-ions can be stored easily between them.
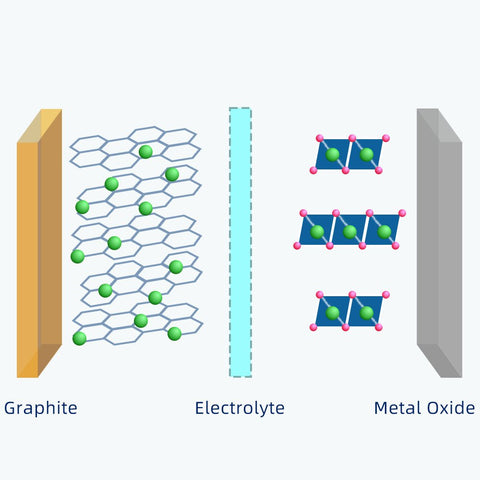
During charging, there are lithium atoms are released from the metal oxide.
Because the lithium atom is highly unstable, it will instantly form a lithium-ion.
The electrolyte between the metal oxide and graphite acts as a guard which allows only lithium-ions to pass through.
After the lithium-ions flow through the electrolyte, they reach the negative electrode and get trapped there.
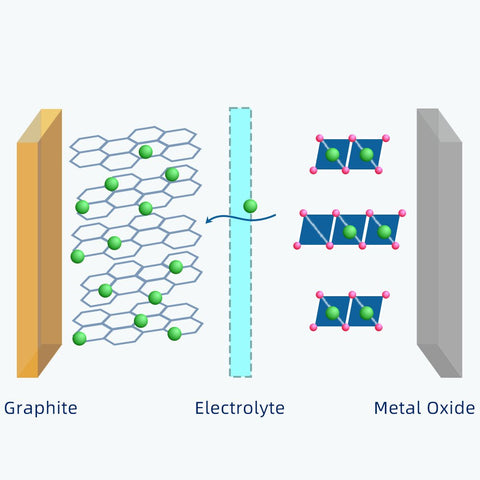
As soon as the power source is removed, this transportation goes in the opposite direction. Those lithium-ions will go back to the metal oxide.
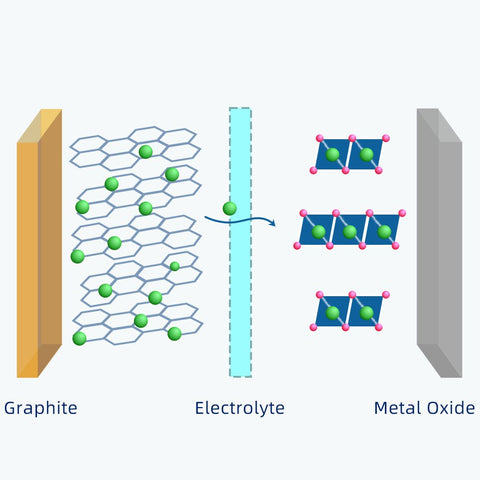
Each lithium atom has only one electron. When a lithium atom transforms into lithium-ion, it will lose the electron.
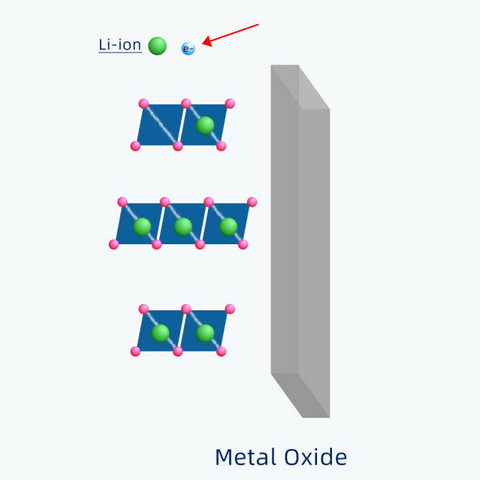
Unlike lithium-ions moving back and forth through electrolytes, electrons move only through the external circuit. That's how electricity is generated.
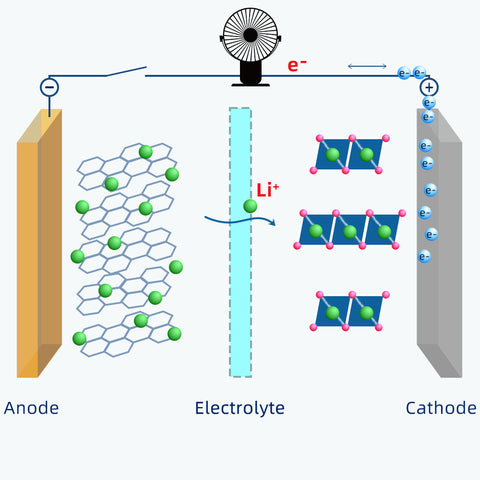
In a practical 18650 cell, the metal oxide is coated onto aluminum foils.
The graphite is coated onto copper foils.

The foils are current collectors here with the positive and negative tabs attached to them respectively.

For safety reasons, a separator sheet is used to prevent contact between positive and negative electrodes.
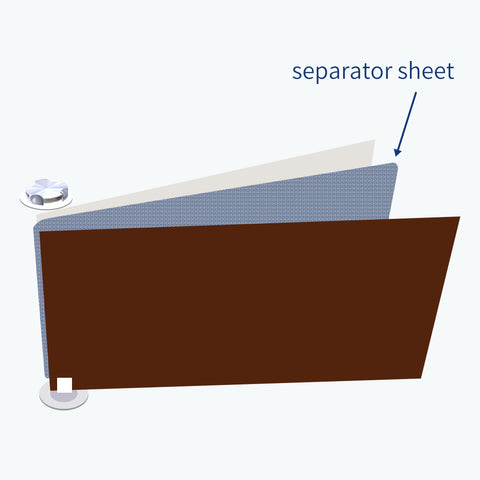
So, there are basically three kinds of sheets. Positive electrode, negative electrode, and separators.
All these three sheets are wound into a cylinder around a center pin, making the cell more compact.

For further protection, they are inserted into a stainless battery case.

Before sealing the case, separators and electrodes are filled completely with electrolyte.
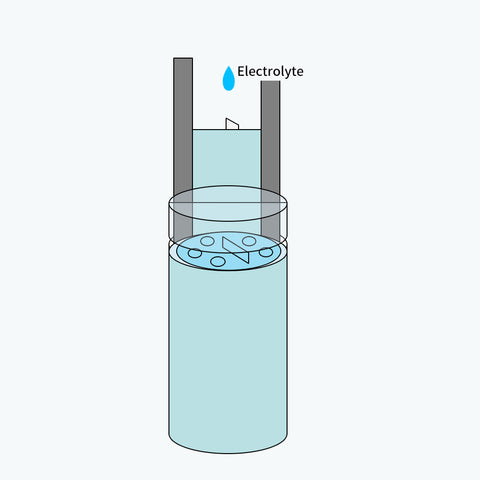
Since the stainless battery case is a metal that conducts electricity, an extra insulating PVC wrapper is needed.
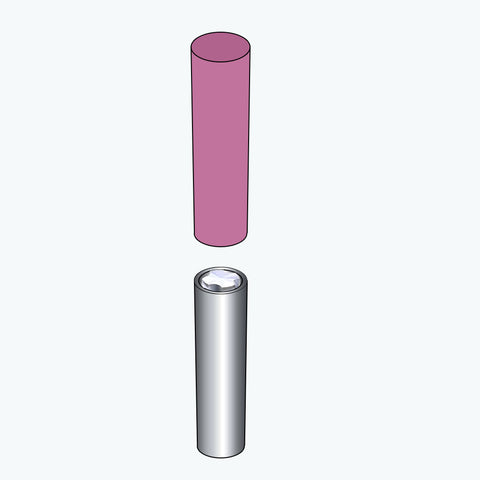
So, there are five major steps of the manufacturing process
Wrapping the battery with a protective cover
.There have been many reported cases about the dangers of lithium batteries.
For example, there have been reports of fires in Tesla cars and exploding power banks in various other applications.
These serious safety issues were almost always caused by an internal short circuits.
An internal short circuit can be triggered by various causes.
It sometimes happens because the separator in a battery melts due to overheating.

Without the protection of the separator, two electrodes could touch each other very easily, causing a short circuit.
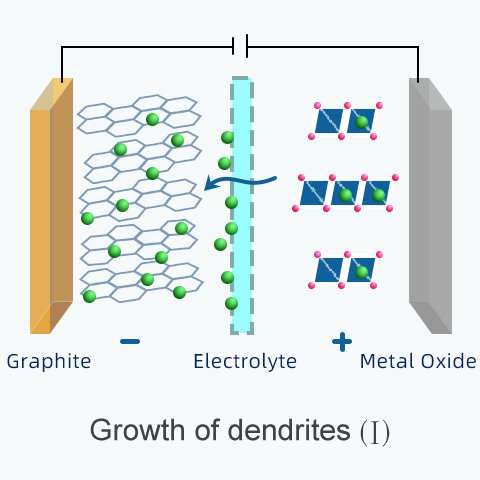
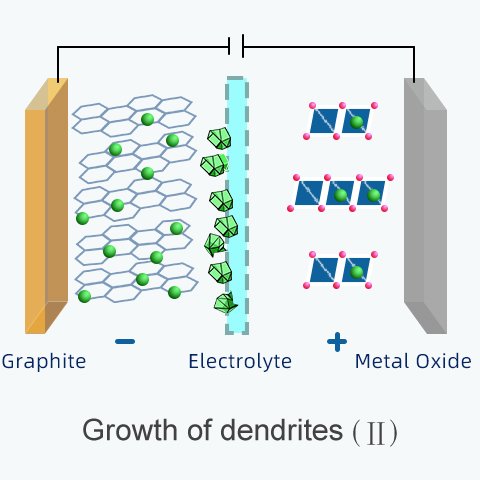
Another cause of internal short circuits is the growth of dendrites.
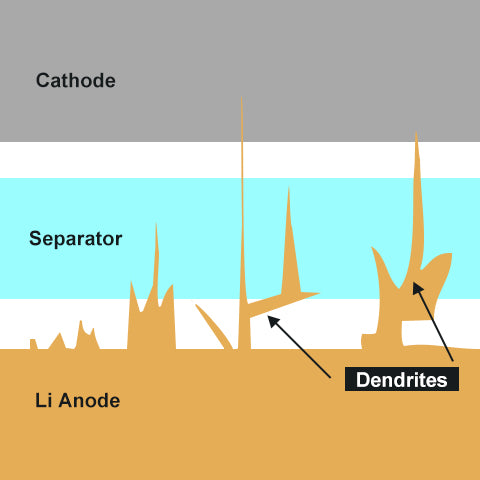
Dendrites (from the Greek dendron meaning "tree"), form on the surface of an electrode.
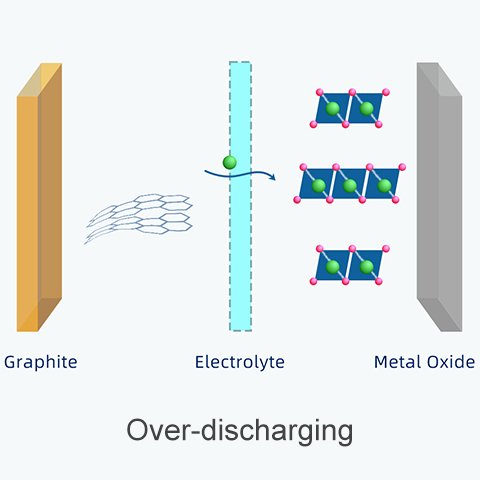
They grow over repeated cycles of overcharging and over-discharging.
(A) Charging: When charging a battery, the lithium-ions move from the cathode to anode.
(B) Overcharging: Numerous ions fill the anode. They are stuck in the graphite and unable to get out. If ions were trapped, electrons cannot be released. That's why the battery capacity is reduced. Since there is limited room in the graphite to accommodate lithium-ions, excess ions will begin to accumulate on the surface with nowhere else to go.
![]()
(C) Discharging: When discharge occurs in the battery, ions will return to the cathode.
(D) Over-discharging: The layered structure has a tendency to collapse as graphite when it contains only a small number of ions. The collapsed structure becomes less able to accommodate ions during the following recharging cycle. This problem makes overcharging more likely to occur.
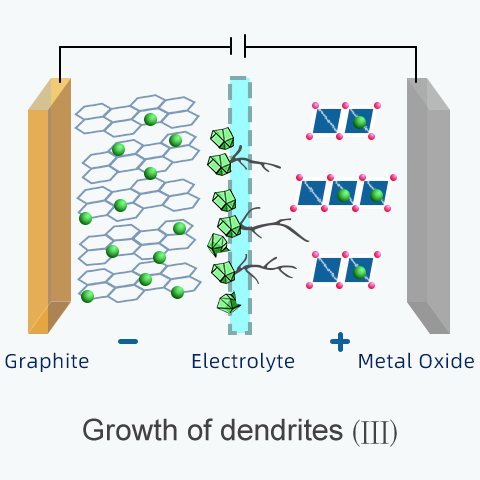
Once the short-circuit happens, the damage is irreversible.
All the battery energy converts into heat. The voltage drops to zero. It won't hold a charge anymore, which leads to the premature replacement of a battery.
In conclusion, maintaining the good conditions of lithium batteries is very important.
Users should minimize the occurrence of dendrites by properly charging and discharging the battery. Making sure the battery is stored at moderate temperature is another practice to keep internal short-circuits from happening.
The battery's positive end is flat.
The battery's positive end is protruding.

Before you are trying to swap out the battery, look at the + pole.
Some devices, will only accept a flat top cell.

As the flat top ones are slightly shorter, a button top will not fit in vaping devices, for example.
Some brands of flashlights like Nitecore & Jetbeam, only work with button-top batteries.

Aside from the size, it's also worth mentioning that NOT all of the button top batteries are protected.
There are opinions like "the button top comes with a protected circuit, and the flat top is unprotected".
This is not true.
Below, we will walk you through the terminologies for protected and unprotected batteries, and how to know which one is protected.
Unprotected battery. With this name, it sounds unsafe and acts like a buyer-repellent.
If that were the case, they will NOT be allowed in the market. So, how come there are so many unprotected cells out there?
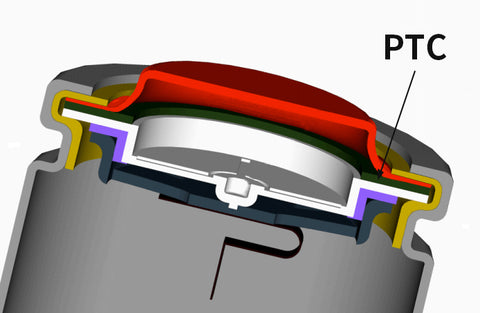
Actually, unprotected battery has the PTC and CID, which means they are safe when properly handled.
PTC thermistor is a small disc that is found at the top of the battery.
When the temperature rises, the resistance increases.
If there is a surge current and the battery heats up significantly, the PTC will increase its resistance to limit the current.
And then the temperature will drop normal, which prevents your battery from overheating.
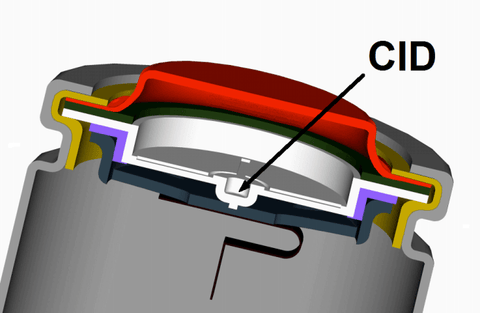
When the battery is overcharged and the pressure inside becomes too high, the CID will disrupt the connection of the positive terminal.
Imagine you are pumping air into a balloon.
After it has reached the optimal level, you are still blowing air into the balloon.
Guess what happens? It burst!
CID, which is like a small hole (the pressure valve), works by releasing the gas through the hole.
So, the "balloon" will slowly leak without causing an explosion.
PTC and CID are built into the majority of 18650 batteries. They prevent the danger of fire and explosion. So, users are not exposed to the potential risks associated with batteries.
However, overcharge and over-discharge can still happen, causing damage to the battery itself.
PCB, the most advanced protection method, helps resolve the issue by detecting voltage above or below safe limits.
With the help of PCB, the battery will disconnect at a certain under, or over-voltage.
It protects the battery against overcharging and over-discharging, assuring longer battery life.
Limit charge voltage, the maximum voltage of 18650 battery voltage, is 4.2V.
When charging the battery, the voltage keeps increasing.
Once the voltage reaches above 4.2V, it can be considered as overcharge.
Cut-off voltage, the lower limit of a 18650 cell varies from 2.75V to 3V, depending on the specification.
It is recommended NOT to discharge the battery lower than 3V, just to be on the safe side.
You may know battery discharges when it connects to a device that needs power.
However, the chemical discharge reaction still happens, albeit relatively slowly, when the battery is not connected to any load.
This phenomenon is called self-discharge.
Self-discharging is not a manufacturing defect.
It occurs to all the batteries.
Although batteries stop connection with any devices when the voltage drops to around 3V, self-discharge will continue to consume the battery's energy.
As mentioned above, rechargeable li-ion batteries is charged and discharged by the movement of li-ions through the electrolyte.
At a very low voltage, the ions cease their movement entirely. That's usually why your device has trouble holding a charge.
You can run a lithium-ion cell totally flat and nothing dangerous will happen.
The thing is, once voltage decreases to the minimum limit, the lithium ions inside are not active anymore, and the cell cannot be recharged.
Sellers and manufacturers may ask you to recharge the battery every 3 months when it's not being used, which can help voltage remain at an optimal level.
Don't panic if you found your battery was the unprotected type when opening the battery compartment.
Modern devices are way smarter with managing power.
Both your device and charger have built-in protection.
They protect the battery against excessive charge and discharge conditions.
That's why unprotected batteries are still being widely used in consumer electronics devices.
Some electronic hobbyists demand "raw" or unprocessed batteries to build their own battery packs. Besides, electronics manufacturers will purchase unprotected batteries from industrial distributors and design the protection circuit into the OEM products.
In all situations, PCB will be added to the device or the battery packs.
So, there's not really any reason to worry about it. The only danger comes from misusing these batteries and causing a short circuit. All you need to do is to use and maintain the device and battery properly.
However, don't ever think about adding PCB on your own. With limited knowledge about electrical engineering, most people don't know what they're handling.
Online sellers should use a pop-up disclaimer and help their customers making an educated purchase decision.
As mentioned above, a protected battery includes PCB protection.
1. Overvoltage: When the battery voltage reaches 4.35V, PCB makes it stop charging.
2. Over-discharging: PCB limits the voltage to 2.5V to prevent the battery from over-discharging.
3. Overcharging: PCB controls the maximum charge to, 3.4Ah (3400mAh, for an 18650 cell), which protects the battery against overcharging.
4. Short circuit: PCB works by preventing the flow of current. This method is kind of like the third one. But this time, it reacts much quicker (usually within 0.007 seconds) to the surge current.
To identify if your battery is protected, check to see if there is a thin ridge under the plastic wrapper on the outside of the cell. This is a characteristic of a protected cell.

With a protected battery, you'll be able to feel the pole connecting the wire to the side of your battery. While the surface of unprotected battery appears smooth.
You can also identify a protected cell as it's generally longer than the unprotected version. This is due to the PCB board attached to the cell bottom.

Finally, the bottom of a protected battery usually indicates its status. It will differ in color from the main body.

If you are looking for a protected 18650 battery, check the spec. Reputable sellers should list this information on the product page.
The specific voltage of a working 18650 battery ranges from 3.2V to 4.2V
The voltage limit setting of PCB is 3V ~ 4.35V.
Unfortunately, this control system is not very precise.
PCB doesn't stop charging until the voltage reaches 4.35V. But the peak voltage of the battery should be 4.2V.
For the sake of longer battery life, don't leave it charging overnight. The advised charging time of one single 18650 cell in a typical charger is between 2 to 3 hours.
There are many dangerous goods on sale. But people are aware of the potential risks of those goods and take caution.
Cars can be dangerous. Therefore, there are driving license and traffic laws.
Excessive alcohol use can be dangerous. Thus, warning signs and education are required.
Most people seeing rechargeable lithium-ion batteries would probably assume they are normal batteries.
Few people know the danger of mishandling an unprotected battery.
Sellers should make customers aware of the correct use and the danger of incorrect use when it comes to lithium batteries or the devices that contain them.
Comments will be approved before showing up.
Email-only promotions.
Directly to your inbox.